
Even, leaving alopecia areata untreated is a legitimate option for many patients. The rate of spontaneous remission makes it difficult to assess the efficacy of a therapy. These autoimmune comorbidities suggest that patients with alopecia areata are at increased risk of developing various T cell-driven inflammatory skin diseases. The nail manifestations consist of fine pitting of the nails, and occasionally, trachyonychia.Īlopecia areata carries associations with several comorbidities, including depression, anxiety, and several autoimmune diseases (thyroid disease, lupus erythematosus, vitiligo, psoriasis, rheumatoid arthritis, inflammatory bowel disease). Patients with nail involvement usually present the most severe forms of hair loss. 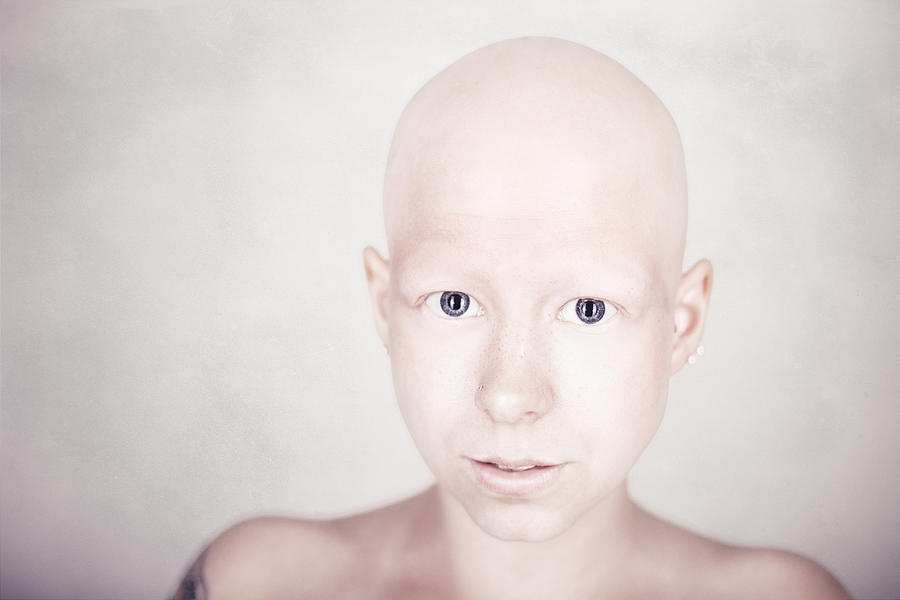
Īlopecia areata can involve the nails, which occurs in about 10 to 15% of cases. Exclamation point hairs can be found at the margins of the lesions during the active phases of the disease. In exclamation-mark hairs, the hair root is narrowed and suggest defective anchoring of the hair within the follicle. Įxclamation-mark hairs are a key characteristic of alopecia areata. Moreover, it may induce complete hair loss on the whole body (alopecia areata universalis) Other variants include ophiasis, in which hair loss affects the occipital region, diffuse forms of alopecia (alopecia incognito) and “sudden graying,” a variant which attacks pigmented hair follicles. The onset is usually fast, and it may progress and induce complete hair loss on the scalp, a condition termed alopecia areata totalis. Although any hair-bearing can be affected, this occurs most commonly on the scalp and beard. It presents as hair loss in well-circumscribed patches of normal-appearing skin. Some triggers have been reported, most commonly emotional or physical stress, vaccines, viral infections, and drugs. 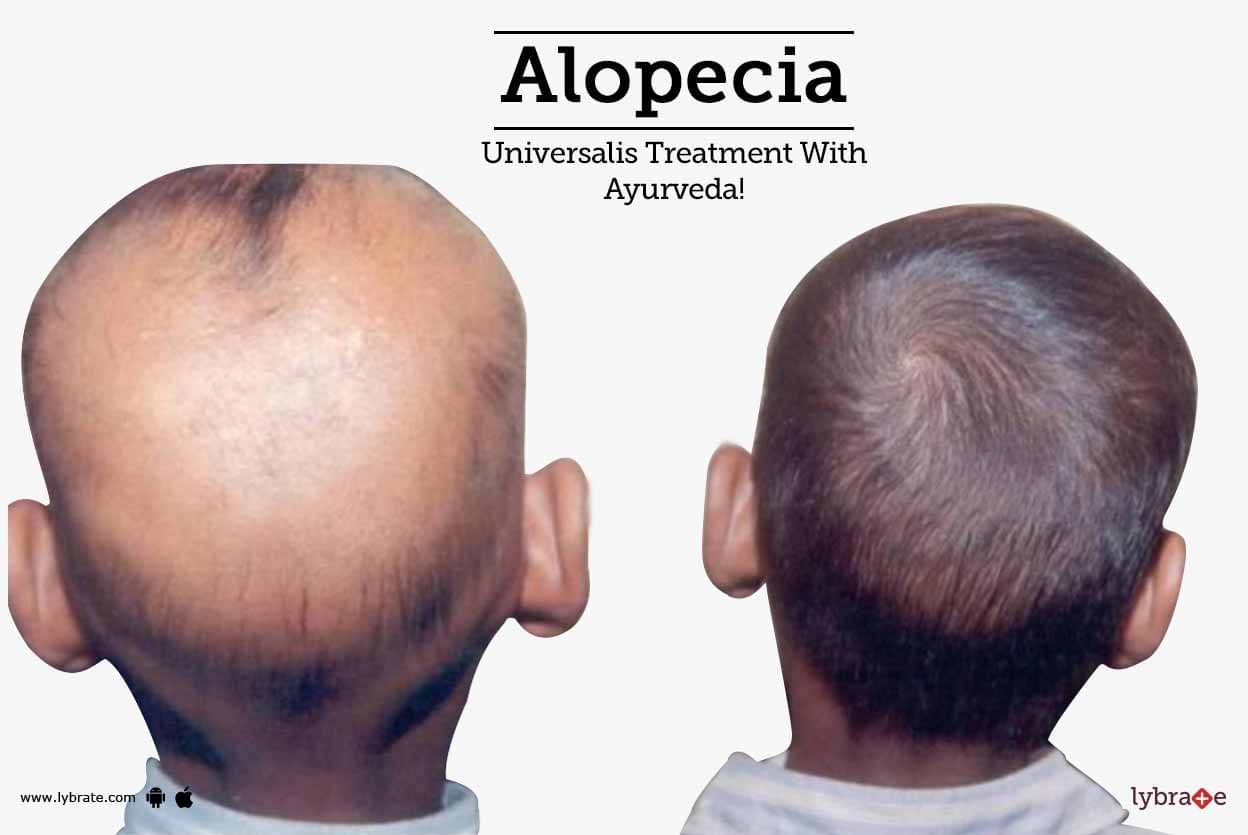
The precise event that precipitates alopecia areata is unknown. Since only anagen hair follicles undergo attack, those autoantigens may generate and then be presented only during anagen. Therefore, it could occur in a genetically predisposed person only when proinflammatory signals (i.e., IFN gamma, substance P) known to upregulate MHC class Ia in human hair-follicle epithelium expose previously unrevealed follicle-associated autoantigens to preexisting autoreactive CD8+ T cells. Presumably, alopecia areata develops in a previously healthy hair follicle because its immune privilege collapses. Thereby, follicles re-enter the anagen phase normally but do not develop beyond the anagen III/IV phase. However, since no destruction of hair-follicle stem cells occurs, the hair follicle retains its capacity to regenerate and continue cycling. Īlopecia areata is a disorder of hair follicle-cycling, where inflammatory cells attack the hair follicle matrix epithelium that is undergoing early cortical differentiation (anagen hair follicles), which are then prematurely induced into the catagen phase. The breakdown of the immune privilege of the hair follicle has been thought to be a significant driver of AA. A significant feature of the hair follicle is its relative immune privilege, this mainly established by suppression of surface molecules required for presenting autoantigens to CD8+ T lymphocytes (i.e., MHC class I) and by the generation of an inhibitory local signaling environment.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
